A team led by Chinese scientists has identified two novel coronavirus-neutralizing antibodies with high specificity from survivors of novel coronavirus pneumonia disease, and demonstrated the therapeutic and preventive potential of one of the neutralizing antibodies in a rhesus monkey model.
The academic journal Nature published the research online in the form of an "accelerated preview" of a novel coronavirus-neutralizing antibody.
It is worth mentioning that this is also the first time since the outbreak of novel coronavirus pneumonia that scientists have reported the results of experiments with neutralizing antibodies against novel coronavirus in a non-human primate model, which has important implications for guiding the design of human clinical trials.
In this study, to find neutralizing antibodies that specifically recognize novel coronaviruses, scientists sampled peripheral blood mononuclear cells from a recovering patient with novel coronavirus pneumonia disease.
They used recombinant novel coronavirus receptor-binding domains (RBDs) as "decoys" and obtained memory B cells specific for these monocytes.
By sequencing and analyzing these B cells, the researchers found two monoclonal antibodies, which they named CA1 and CB6, for subsequent studies.
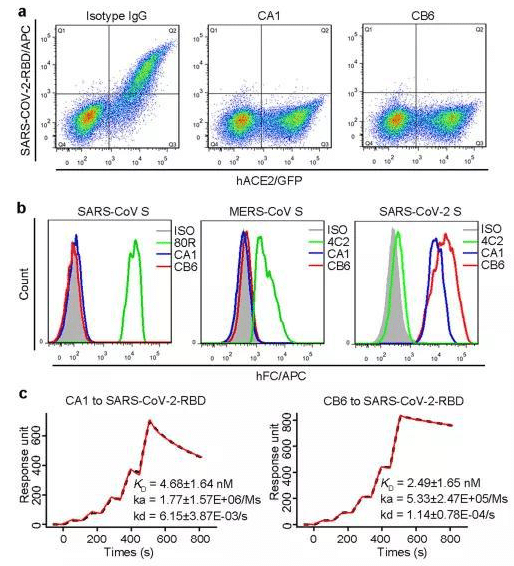
▲The researchers identified two monoclonal antibodies that specifically recognize the novel coronavirus RBD and block the novel coronavirus from binding to the ACE2 receptor.
Cellular experiments showed that both monoclonal antibodies prevented the novel coronavirus RBD from binding to the human ACE2 receptor on the cell surface. In subsequent other cellular experiments, both antibodies also showed good neutralizing activity, while CB6 showed a stronger neutralizing ability.
Scientists then examined the therapeutic and preventive effects of CB6 antibodies in animals.
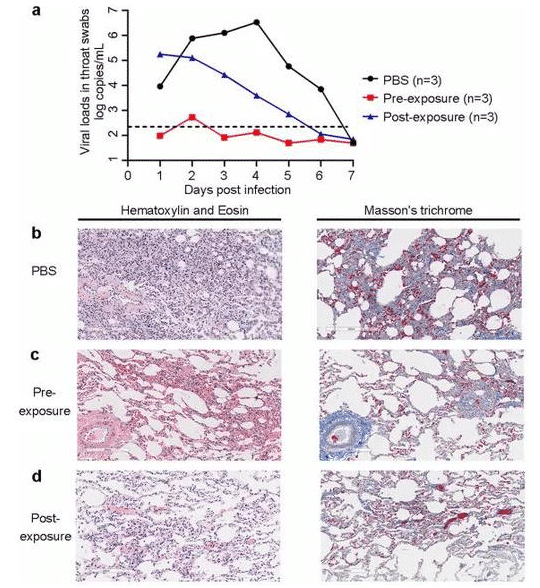
Specifically, the scientists used nine rhesus monkeys, divided them evenly into three groups (treatment group, prevention group, and control group), and infected them with the novel coronavirus to observe the effect of neutralizing antibodies.
Of these, the treatment group was injected with CB6 antibodies on the first and third days after viral infection, the prevention group was injected with CB6 antibodies on the day before viral infection, and the control group was injected with saline (PBS) placebo.
▲CB6 reduced novel coronavirus load in both the treatment (blue) and prevention (red) groups. In addition, this antibody improves the pathological characteristics of the animal.
Results from non-human primate experiments showed a definite therapeutic effect in monkeys injected with CB6 antibodies on day 4 of novel coronavirus infection with a significant reduction in viral titers in vivo.
In the prevention group, the CB6 antibody injection also had a significant preventive effect, resulting in significant protection for the animals. The results of the pharyngeal swab showed that the animals in the prevention group, had lower levels of the virus in their bodies.
Pulmonary pathological analysis of these monkeys also showed that CB6 reduced the associated lung damage caused by novel coronaviruses.
The researchers noted that the antibody not only inhibited the titers of the novel coronavirus, but also improved infection-related lung pathology within the prevention and treatment groups.
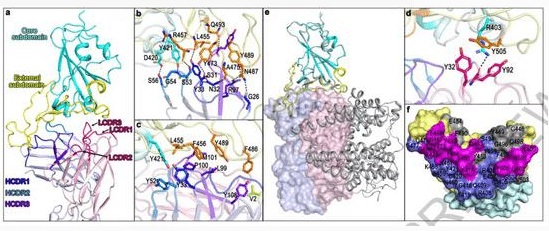
The scientists also analyzed the crystal structure of the neutralizing antibody in order to understand its specific mechanism of action.
Resolution revealed that the CB6 antibody recognizes the very site in the novel coronavirus RBD that binds to the human cell surface receptor ACE2, which can interfere with virus-receptor interactions.
This also mechanistically explains why this antibody exhibits potential for treatment and prevention.
▲CB6 can compete with ACE2, thereby interfering with viral-receptor interactions.
The study was jointly conducted by the Institute of Microbiology of the Chinese Academy of Sciences, Wuhan Institute of Viruses of the Chinese Academy of Sciences, Beijing Ditan Hospital, the Fifth Medical Center of the People's Liberation Army General Hospital, the Institute of Viral Diseases of the National Center for Disease Prevention and Control, and Junshi Biosciences.
Based on the potential of CB6, Junshi Biosciences has also partnered with Eli Lilly and Company to submit clinical trial applications and initiate human clinical studies in China and the United States in the second quarter of this year.
Dr. Yan Jinghua, a researcher at the Institute of Microbiology, Chinese Academy of Sciences, one of the study leaders, concluded in a press release from Junshi Biosciences, "This study reports two monoclonal antibodies with good neutralizing activity. The antibody code-named CB6 showed a stronger blocking ability in vitro, not only has an affinity 100-200 times higher than that of the host cell receptor, but also has a high degree of overlap with the viral binding region, making the possibility of viral binding to the host cell greatly reduced. At the same time, we have also demonstrated the therapeutic and prophylactic power of CB6 antibodies in non-human primate experiments, indicating that this drug candidate against novel coronaviruses is very promising and we very much look forward to its further validation in clinical trials."
Dr. Feng Hui, COO of Junshi Biosciences, added, "Based on the company's years of experience in full lifecycle technology platforms for antibody development, we are conducting multiplex parallel development of candidate antibodies screened by CAS Microbiology. As published in Nature, one of the candidate antibodies exhibited strong neutralizing activity against a new coronavirus. We look forward to working with Eli Lilly to advance the clinical application for this antibody in the second quarter, and Junshi Biosciences will continue to step up its research and development efforts to leverage its leadership in innovative biologics and contribute to the fight against epidemics in China and globally."
Special Report: Fighting The New Coronavirus