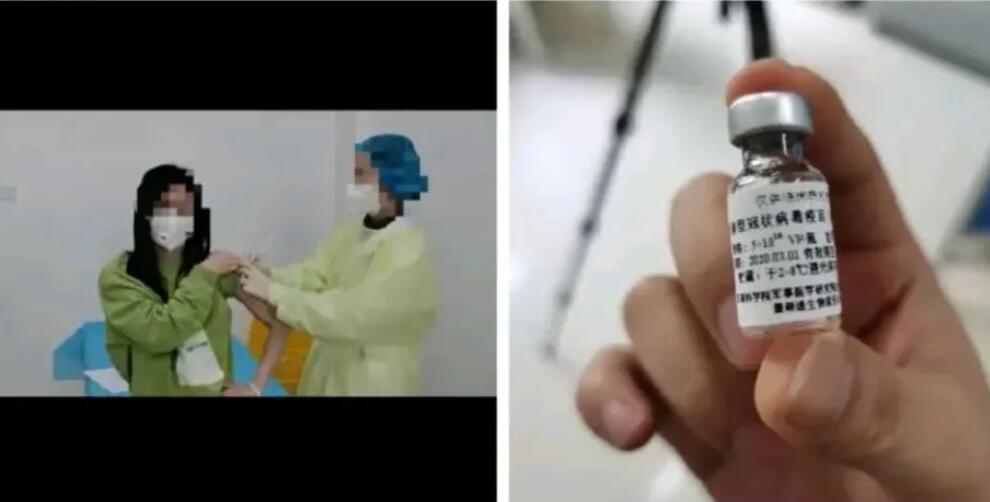
A new coronavirus vaccine developed in China has been injected into humans. There are 3 groups of volunteers with each group having 36 people. They will be observed in isolation for 14 days after injection, according to the Science and Technology Daily.
Organizers of the trial are the Institute of Bioengineering of the Academy of Medical Sciences of the Academy of Military Sciences and Cansino Biotech.
The purpose of the test is to test and evaluate the safety and effectiveness of the new recombinant coronavirus vaccine (adenovirus vector).
This vaccine was constructed using genetic engineering methods, using replication-defective human type 5 adenovirus as a vector, and expressing the new coronavirus S antigen.
Chen Wei, the project leader, a member of the Chinese Academy of Engineering and a researcher at the Military Medical Research Institute, explained its principle in this way: on the premise of "learning" the virus, "surgery" the virus, using flowers and trees to transform a carrier we need The virus is injected into the body to produce immunity.
There are not many volunteers required for Phase I trials. They are limited to residents of Wuhan, and residents of Wuchang, Hongshan, and Donghu Scenic Areas are preferred.
Volunteers will be divided into three groups of low-dose, medium-dose and high-dose groups, with 36 people in each group.
After screening and physical examination, eligible volunteers can be vaccinated. The next 14 days are the observation period for centralized isolation.
Within six months after the vaccination, the medical team will periodically follow up with the volunteers several times to see if they have any adverse reactions and whether anti-S protein-specific antibodies are produced in the body.
Special Report: Fighting The New Coronavirus